|
Note 2: Born-Haber cycles where the charge on the negative ion is more negative than -1Lattice Energy is a type of potential energy that may be defined in two ways. Youll also need 2 x atH for the Cl 2(g) to be turned into 2Cl (g). If forming CaCl 2, then youll need 2x EAH to turn two 2Cl (g) into 2Cl-(g) in order to form CaCl 2. Some Born-Haber cycles will require multiples of the relevant energy change, e.g.As implied in the definition, this process will always be exothermic, and thus the value for lattice energy will be negative. The other definition says that lattice energy is the reverse process, meaning it is the energy released when gaseous ions bind to form an ionic solid. This definition causes the value for the lattice energy to always be positive, since this will always be an endothermic reaction.
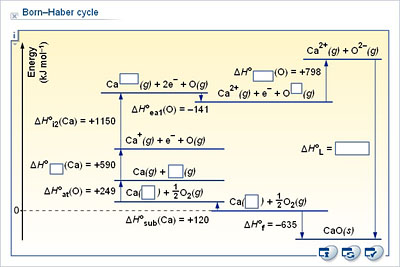  In general, ionization energy increases across the periodic table from left to right, and decreases from top to bottom. This process always requires an input of energy, and thus will always have a positive value. Ionization Energy is the energy required to remove an electron from a neutral atom or an ion. Some require such high temperatures that they decompose before they can reach a melting and/or boiling point.There are several important concept to understand before the Born-Haber Cycle can be applied to determine the lattice energy of an ionic solid ionization energy, electron affinity, dissociation energy, sublimation energy, heat of formation, and Hess's Law. It is this that causes ionic solids to have such high melting and boiling points. In general, ionization energy increases across the periodic table from left to right, and decreases from top to bottom. This process always requires an input of energy, and thus will always have a positive value. Ionization Energy is the energy required to remove an electron from a neutral atom or an ion. Some require such high temperatures that they decompose before they can reach a melting and/or boiling point.There are several important concept to understand before the Born-Haber Cycle can be applied to determine the lattice energy of an ionic solid ionization energy, electron affinity, dissociation energy, sublimation energy, heat of formation, and Hess's Law. It is this that causes ionic solids to have such high melting and boiling points.
Therefore, when used in calculating the lattice energy, we must remember to subtract the electron affinity, not add it. Usually, energy released would have a negative value, but due to the definition of electron affinity, it is written as a positive value in most tables. Electron Affinity is the energy released when an electron is added to a neutral atom or an ion. Old english font for mac freeTherefore, the change in energy is always positive. The dissociation of a compound is always an endothermic process, meaning it will always require an input of energy. Dissociation energy is the energy required to break apart a compound. Does usb to vga work for macThis may be positive or negative, depending on the atoms involved and how they interact. The heat of formation is the change in energy when forming a compound from its elements. It may also be referred to as the energy of atomization. This is an input of energy, and thus has a positive value. Sublimation energy is the energy required to cause a change of phase from solid to gas, bypassing the liquid phase. The Born-Haber Cycle is essentially Hess's Law applied to an ionic solid.
0 Comments
Leave a Reply. |
AuthorBrandon ArchivesCategories |
 RSS Feed
RSS Feed
